Indicators
What indicators are, why they are useful and why each is good at doing a particular thing
|
|
Indicators are very important in the chemistry laboratory! While universal indicator is useful to determine the exact pH of substance, other indicators, that have a single colour change are used to determine a reactions end-point or if something has gone beyond a specific pH.
It is for this that: --> Universal indicator is NOT the most useful indicator -->Different indicators are useful for different types of reaction --> Liquid, indicators can change the pH they are testing! --> Liquid indicators cannot be used to test the pH of reactions already involving strong colours --> Sometimes an electric pH meter is more useful than an indicator --> An indicator may not be needed to determine the reaction end-point! |
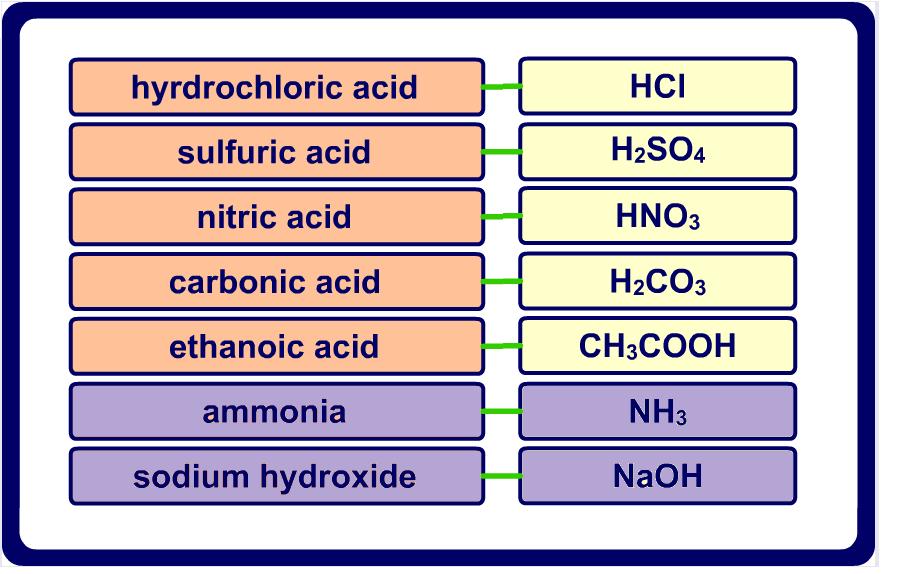
Acids & Bases
What acids and bases are and how they react with each other
|
|
|
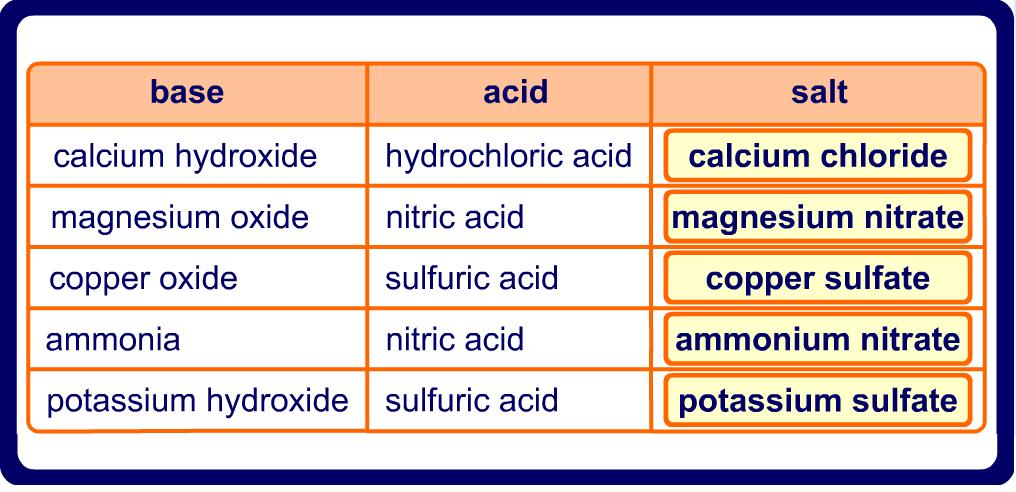
Naming Salts
Salts are made when a metal and a non-metal bond together in an ionic bond
|
See the worksheet below for easy instructions on how to name salts
Salts have many uses in industry - they are also used to give fireworks their beautiful colours.
Each salt burns a bright and unique colour, see the video to the right where balloons with different salts inside are burst to show the flame colour of the salt. |
Examples of the salts made in acid-base reactions are shown below. Take care not to forget that all of these reactions make other products too!
|